Unlocking the Secrets of Molecules: A Visual Guide to Understanding Sp Hybridization
Understanding the behavior of molecules is a crucial aspect of chemistry, and at its core is the concept of hybridization. Hybridization is a theoretical model that helps us comprehend how atomic orbitals combine to form molecular orbitals. One of the key types of hybridization is sp hybridization, a fundamental concept that is often misunderstood or overlooked, especially in undergraduate chemistry courses. This article will delve into the basics of sp hybridization, its characteristics, and how it affects the geometry of molecules.
Hybridization is a crucial concept in chemistry, and a deep understanding of it can be the difference between a good solid hold on the subject and a muddled understanding. When chemists describe the bond angle for a molecule, this can be a sign of understanding the hybridization that is happening within the molecule. The key to comprehension is looking at the linear geometry.
**What is Sp Hybridization?**
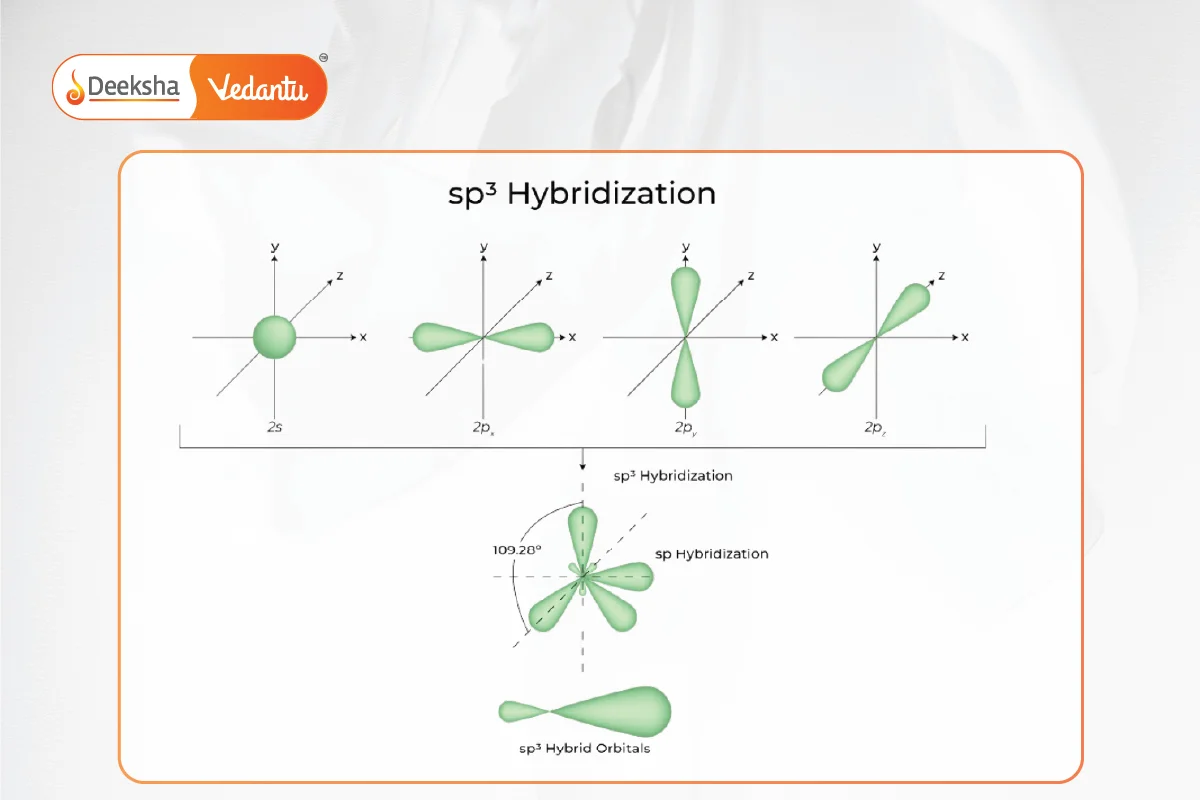
Hybridization is a concept in chemistry that describes the mixing of different atomic orbitals to form new hybrid orbitals. In the case of sp hybridization, one s orbital and one p orbital are combined to form two equivalent sp orbitals. This type of hybridization typically occurs in molecules with a triple bond, for instance C≡C arrangements. These orbitals have a 50% s and 50% p nature, giving them a different shape and orientation compared to p orbitals.
When sp hybrid orbitals are created, the molecule typically adopts a linear geometry where the two sp orbitals are arranged in a straight line. This is because the sp orbitals lie along the nodal plane, which is an imaginary plane that separates the molecule into two equal halves.
An example of a molecule that exhibits sp hybridization is carbon in acetylene (C2H2), a compound consisting of two carbon atoms triple-bonded to each other. The two carbon atoms are linear and are denoted as sp-hybridized. On the other hand, the carbon atoms in CO2 are sp hybridized whereas carbon atoms in CH4 are not, existing in an sp^3 hybridization.
**Properties of Sp Hybridization**
Sp-hybridized molecules exhibit several distinct properties:
* **Linear Geometry**: As mentioned earlier, sp-hybridized molecules typically adopt a linear geometry due to the directional nature of the hybrid orbitals.
* **Bond Angle**: Since sp hybrid orbitals are directed along the bond axis, the bond angle in sp-hybridized molecules is always 180°.
* **Multiple Bonds**: Sp hybridization often results in multiple bonds between atoms, such as triple bonds.
* **Molecular Stability**: The stability of sp-hybridized molecules can be due to the overlap between sp orbitals and the bonding between them.
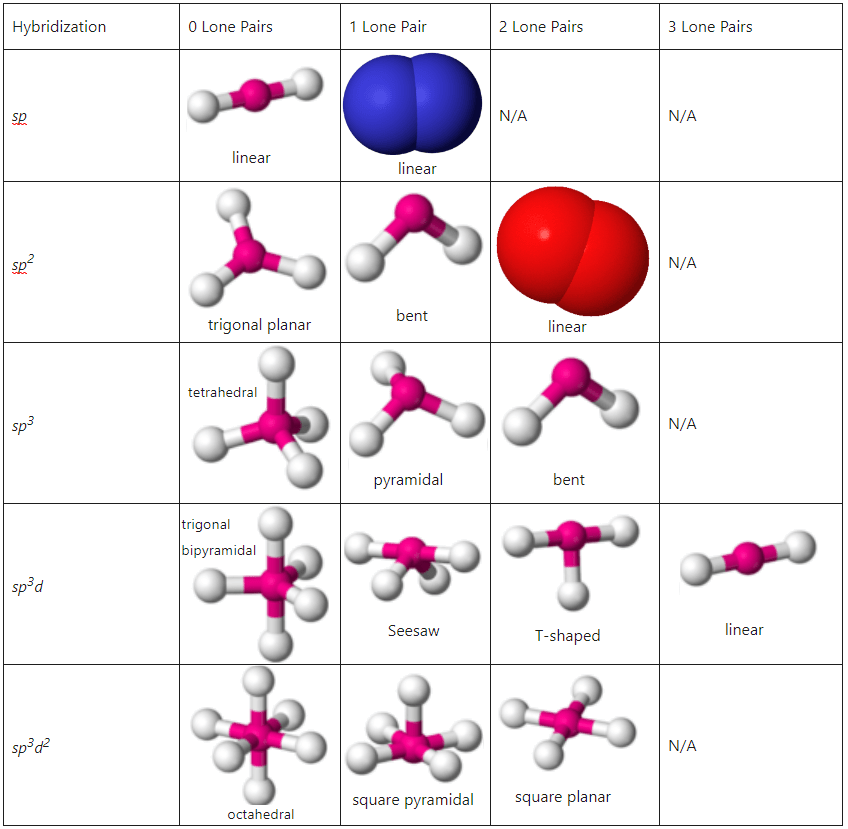
**Types of Hybridization**
There are several types of hybridization, each with specific characteristics:
- sp^3 hybridization: Formation of four equivalent sp^3 orbitals from one s and three p orbitals, found in CH4 and other tetrahedral molecules.
- sp^2 hybridization: Formation of three equivalent sp^2 orbitals from one s and two p orbitals, found in planar molecules such as ethene (C2H4).
- sp hybridization: Formation of two equivalent sp orbitals from one s and one p orbital, found in linear molecules like CO2.
**In Conclusion**
Understanding sp hybridization is crucial in explaining the properties and behaviors of molecules, especially in predicting molecular geometry and orbital orientation. It is not only an essential concept in inorganic and organic chemistry but also biochemistry, as the bonding of atoms in larger molecules relies on these principles.